Thermodynamics is the branch of engineering science concerned with transfer of heat and its conversion to or from mechanical energy in plant and machinery. Common examples are: steam and gas turbines, internal combustion engines, reciprocating and rotary compressors, steam boilers, condensers and heat exchangers, all of which contain one or more working fluids. Thermodynamics is also an important aspect of chemical engineering.
Fluids are contained within thermodynamic systems with defined boundaries and can be present in the gas, vapour and liquid phase within one system. Where all fluid remains within the boundary of a system it is called a closed system. Where fluid enters and leaves the system it is called an open system.
Engineering thermodynamics focuses heavily on the study of non-flow processes in closed systems and steady-flow processes in open systems.
A thermodynamic cycle is a repeatable combination of linked processes in which the state of the fluid is identical at the start and end points of the cycle. Cycles can be open or closed.
The state of a fluid in a system is defined by its thermodynamic properties. We are familiar with the basic properties of pressure \((p)\), temperature \((T)\) and volume \((V)\). For practical applications in thermodynamics we generally use specific volume \((v)\), i.e. volume per unit mass*, as a property, being the inverse of density.
*the advantage of expressing properties in terms per unit mass is explained below.
Two properties alone define the state of a fluid in a closed system. Fluid in a non-flow, closed system is said to be in equilibrium when properties are uniform and stable throughout the system.
In most cases pressure and temperature of a fluid in a system are directly measurable. Other useful derived properties are: internal energy \((U)\), enthalpy \((H)\) and entropy \((S)\), which are defined in subsequent tutorials.
Thermodynamic processes involve changes of state of fluids brought about by transfers of heat energy \((Q)\) and work energy \((W)\). The sign conventions for \(Q\) and \(W\) are:
There are several ways of expressing the First Law, which is based on confirmation by universal observation and experiment. We choose the following expression.
When a closed system is taken through a cycle the net work delivered to the surroundings is equal to the net heat taken from the surroundings.
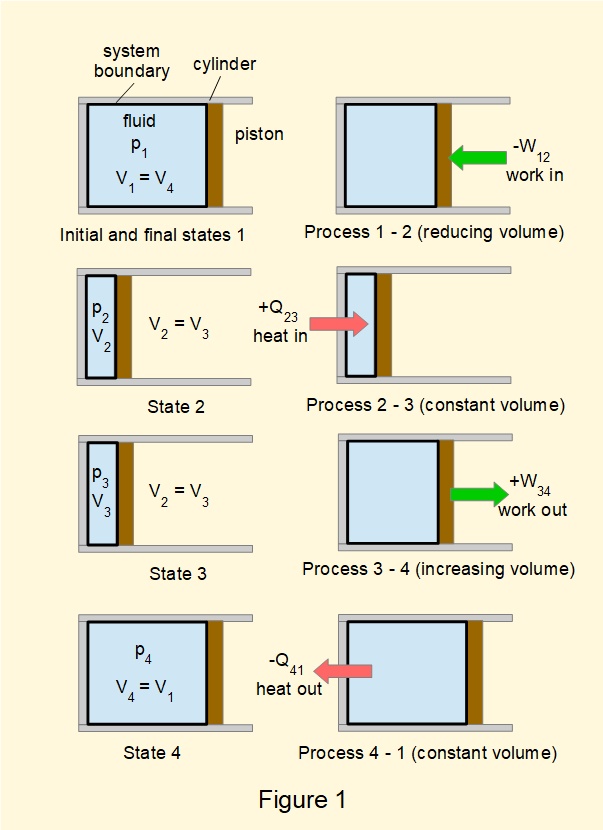
Consider the thermodynamic cycle* for a closed system containing a gas illustrated in Figure 1. We call such cycles that produce mechanical work from input of heat heat engines. Figure 2 illustrates the cycle in plot of pressure \(p\) against volume \(V\) known as a \(pV\) diagram.
(* this cycle is a version of the Otto Cycle which is a theoretical basis for gasoline fuelled internal combustion engines.)

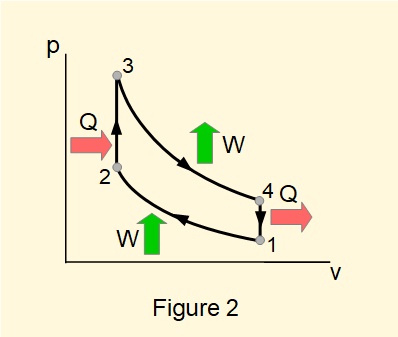
Net amounts of heat and work transferred over the complete cycle are:
\(Q_{net}=Q_{23}-Q_{41}=\Delta{Q}\) and \(W_{net}=W_{34}-W_{12}=\Delta{W}\)
Applying the First Law of Thermodynamics stated above for a cycle in a closed system:
\(Q_{net}=W_{net}\) or \((\Delta{Q}-\Delta{W})=0\)
giving \((Q_{23}-Q_{41})=(W_{34}-W_{12})\)
This example illustrates that the first law is essentially a statement reflecting the principle of conservation of energy in the sense that returning a fluid in a closed system to its original equilibrium state results in neither a net gain nor loss of energy to the surroundings.
We have stated no requirements for this process to ensure that \(p_1\) and \(V_1\) are the same after completing one cycle. The next tutorial addresses this question using the concept of reversibility.
Figure 3 illustrates another interpretation of the First Law introducing the property of internal energy \(U\). The statement is as follows:
There exists a property of a fluid in a closed system (called internal energy) such that a change in its value is equal to the difference between the heat supplied and the work done during any change of state.
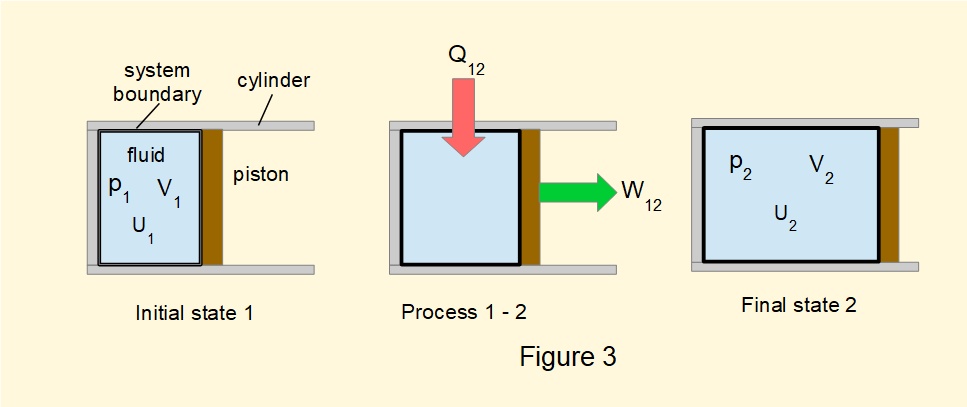
Designating \(U_1\) the value of \(U\) at the start of the change of state and \(U_2\) the value at the end of the change this version of the First Law is expressed as.
\(Q_{12}-W_{12}=(U_2-U_1)\) -------- (1)
Equation (1) is the general energy equation for a closed system.
Internal energy is the random molecular energy of the fluid. It is quantified in thermodynamic applications as the difference \({\Delta}U\) relative to a reference value.
Fluids in thermodynamic systems can also possess kinetic energy \((KE)\) and potential energy \((PE)\) which are important in thermodynamic analysis of flow systems. We use \(E\) to denote the sum of all energy within a fluid, i.e. \(E = (U + KE + PE)\).
Units in thermodynamics
SI units are used in these tutorials, based on mass (kg), length (m) and time (seconds). The table below lists units and symbols for the principal physical quantities and properties.
Quantities \(u, v, h, s\) are expressed per unit mass, indicated by lower case symbols. This allows multiplication of general formulae or equations by whatever mass or mass flow rate applies in a particular application.
For convenience of scale the following deviations from basic SI units are generally adopted:
Using kPa and kJ together in calculations eliminates the need for a conversion factor but in engineering practice operating pressures are typically stated in bars.
| heat energy | \(Q\) | kJ |
| work energy | \(W\) | kJ |
| absolute pressure | \(p\) | bar or kPa |
| absolute thermodynamic temperature | \(T\) | °K |
| thermodynamic temperature | \(t\) | °C |
| temperature (arbitrary scale) | \({\theta}\) | no specific unit |
| density | \({\rho}\) | kg/m3 |
| specific volume | \(v\) | m3/kg |
| specific internal energy | \(u\) | kJ/kg |
| specific enthalpy | \(h\) | kJ/kg |
| specific entropy | \(s\) | kJ/kg°K |
| specific heats | \(c_p\), \(c_v\) | kJ/kg°K |
Next: Processes in closed systems
I welcome feedback at: