Reversible and irreversible processes, work done
Constant volume, constant pressure, polytropic, adiabatic and isothermal processes
In the previous tutorial we introduced the first law of thermodynamics for transfers of heat and work to and from closed systems in terms of the energy equation:
\({\Delta}Q-{\Delta}W={\Delta}U\)
In this tutorial we develop the concepts of reversible and irreversible processes and derive energy equations for specific processes in closed systems.
Reversible process for transfer of work and heat
A reversible process is one where the system is conceived to pass through a sequence of equilibrium states in each of which all properties are uniform throughout the system. Figure 1 illustrates such a system containing an expanding gas in a cylinder producing work \(W\) on a piston.
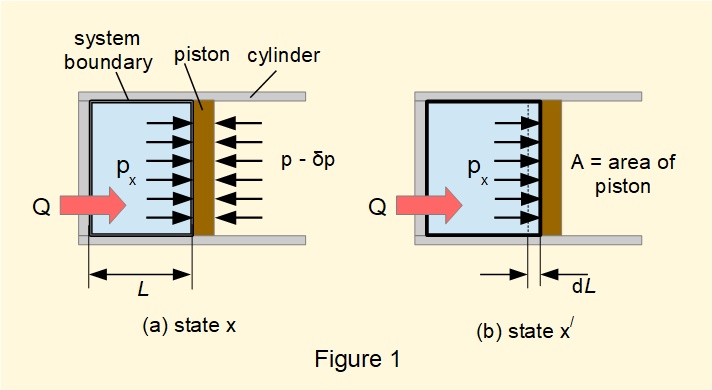
In Figure 1(a) state x is a random equilibrium position in the expansion. Pressure in the cylinder is assumed to remain constant between state x and state x/ at (b) over a very small outward movement of the piston \(dL\) made possible by an infinitesimal difference in pressure \({\delta}p\) on the opposite face of the piston.
Heat transfer is also conceived through a series of equilibrium states where the temperature difference \({\delta}T\) between the system and the heat source or sink is infinitesimally small.
These processes are reversible because by reversing the direction of \({\delta}p\) and \({\delta}T\) they can proceed in the opposite direction back to the initial state.
Work done during a reversible non-flow process
The work done by the reversible process illustrated in Figure 1 between two defined states 1 and 2 is derived as follows.
The force of the gas on the piston \(F=p.A\)
Thus work done over distance dL is: \(dW = F.dL = p.A.dL = p.dV\) where \(dV\) is the corresponding change of volume.
In mathematical terms the relationship between two properties in a reversible process is a continuous function. Thus the total work \(W\) delivered between states 1 and 2 can be expressed as:
\(\large W = \int ^2_1 p.dV \)
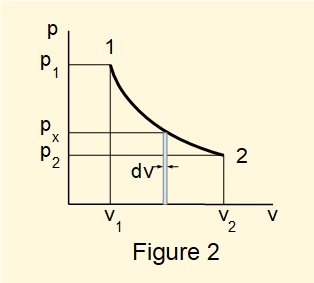
Figure 2 illustrates a reversible process from state 1 to state 2 on a \(pV\) diagram. This type of diagram plotting two properties of the working fluid is frequently used in thermodynamics.
\(\large\int ^2_1 p.dV \) representing the work done is the area under the curve between states 1 and 2.
Sources of irreversibility
In practice it is not physically possible to achieve total reversibility. However, it is a very useful basis for analysing thermodynamic processes. Real processes produce turbulence in the fluid and the associated viscous friction increases internal energy with a consequential reduction in energy available for work. This effect is not reversible.
It is also the case that heat transfer to and from the surroundings through a boundary wall requires a temperature gradient contrary to the criterion of an infinitesimal temperature difference.
Irreversibilities are considered further in a future tutorial dealing with the property entropy.
Figure 3 below shows the convention for illustrating an irreversible process between two states using a dashed line on a \(pv\) diagram.
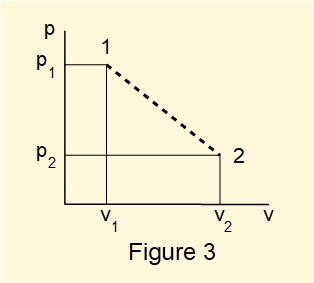
The dashed line does not define the state of the system at any point between states 1 and 2 and the area below the line does not represent the work delivered by the process. Generally, the state of a fluid undergoing an irreversible process cannot be determined from theory. However certain methods and assumptions can provide good approximations which we will use in subsequent tutorials.
Application of the energy equation
We now apply the energy equation of the First Law \(({\Delta}Q-{\Delta}W={\Delta}U)\) to the following specific processes .
- constant volume process
- constant pressure process
- polytropic process
- adiabatic process
- isothermal process
Constant volume process
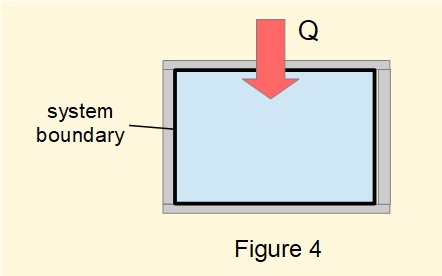
Figure 4 shows a rigid vessel containing a fixed volume of gas. It is physically impossible to obtain work from this system*. By the First Law the process of transferring heat to or from the system from equilibrium state 1 to equilibrium state 2 gives:
\(Q-W=(U_2-U_1)\)
But as \(W=0\) it follows that the energy equation for a constant volume process is:
\(Q=(U_2-U_1)\)
This equation applies to all constant volume processes whether reversible or irreversible because \((U_2-U_1)\) depends only on the end states.
* We can supply work to this system by what is called paddle work, which churns the gas, thus increasing its internal energy giving \(Q=0\) and \(-W=(U_2-U_1)\).
Constant pressure process
(1) Reversible process
Figure 5 shows a closed system containing a gas within a cylinder and piston identical to Figure 1 except that the cylinder sits vertically and a mass \(M\) exerts a constant external force \(F=m.g\) on the piston throughout the process from initial equilibrium state 1 to final equilibrium state 2. A quantity of heat \(Q\) is transferred to the system from an external source which expands the gas through an infinite series of equilibrium states from volume \(V_1\) to \(V_2\) at constant pressure \(p\) equal to constant pressure \(p_{ext}\) acting on the piston. A corresponding process in reverse from state 2 to state 1 can be performed by transferring heat from the system under the same condition of constant pressure \(p\).
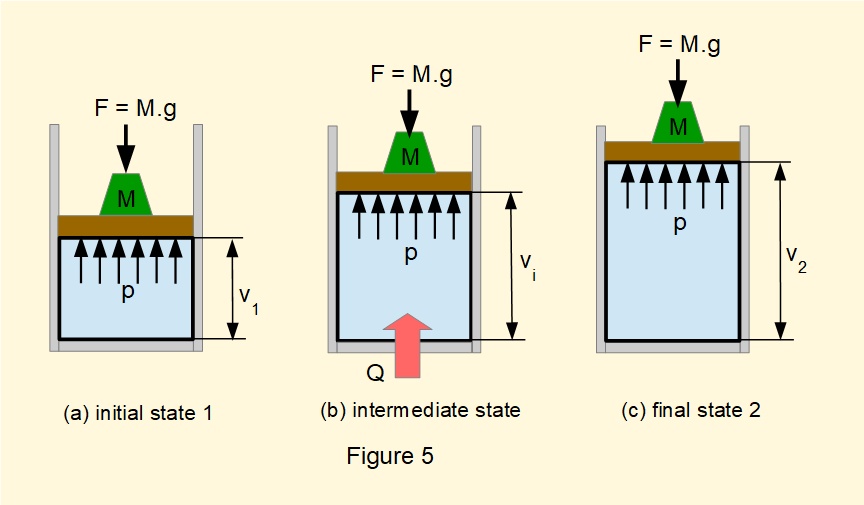
Applying the first law directly to this process: \(Q-W=(U_2-U_1)\)
If the process is reversible: \(dW=p.dV\) thus \(dQ-p.dV=dU\) -------- (1)
Giving by integration: \(Q-p(V_2-V_1)=(U_2-U_1)\)
(2) Irreversible process
In an irreversible constant pressure process only the external pressure on the piston \(p_{ext}\) is constant. Rapid expansion of gas inside the cylinder produces turbulence and viscous friction.
In this case the work done is determined by the external pressure \(p_{ext}\) such that:
\(W=p_{ext}(V_2-V_1)\) and the energy equation becomes:
\(Q-p_{ext}(V_2-V_1)=(U_2-U_1)\)
Definition of enthalpy
In the reversible constant pressure process the pressure of the gas \(p\) is constant thus:
\(d(p.V)=(p.dV+V.dp) = p.dV\) and equation (1) gives:
\(dQ = du + d(p.V)\)
which by the rule for adding differentials gives: \(dQ = d(u + p.V)\)
The quantity \((U + p.V)\), which is a property of the fluid, is called enthalpy \( H\) with units \(kJ\). Enthalpy is an extremely useful property frequently used in analysis of thermodynamic processes and is normally stated as specific enthalpy \(h\) with units \(\large\tfrac{kJ}{kg}\). In common with internal energy values of enthalpy are relative to a reference value.
Thus the energy equation for reversible constant pressure process can be stated as: \(dQ=dH\)
giving by integration: \(Q = (H_2-H_1)\)
Polytropic process
Constant volume and constant pressure processes are limiting examples of a more general process where both pressure and volume vary during expansion and compression.
Many such processes can be represented by an equation of the form:
\(p.v^n=c\) where \(c\) is a constant and \( n\) is the index of expansion.
Such processes are called polytropic.
(from this point we use lower case form for properties, denoting per unit mass)
The \({pv}\) diagram, illustrated in Figure 6, represents states of the system for two reversible polytropic processes: (a) expansion from state 1 to state 2, and (b) compression from state 1 to state 3 along the same curve \(p.v^n=c\). Areas under sections of the curve \(1{\rarr}2\) and \(1{\rarr} 3\) represent respectively work extracted during expansion and work input during compression.
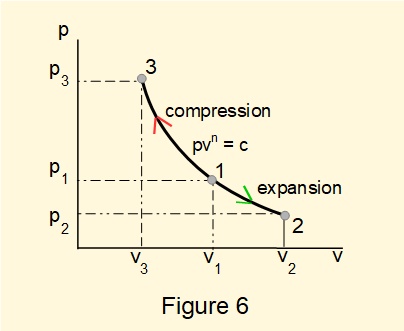
For a reversible polytropic process: \(p_1.{v_1}^n=p_2.{v_2}^n=p.v^n\) giving: \(p=\dfrac{p_1.{v_1}^n}{v^n}\)
thus: \(W=\int_1^2{p.dv}=p_1{v_1}^n\int_1^2\dfrac{dv}{v^n}\) giving: \( {\large W} = {\Large(p_2.v_2-p_1.v_1)\over(1-n)} \)
and the energy equation for a reversible polytropic process is:
\( {\large Q} - {\Large(p_2.v_2-p_1.v_1)\over(1-n)} = {\large (u_2 - u_1)} \)
Adiabatic process
A process where no heat crosses the boundaries of a system is called an adiabatic process and represents a system completely thermally insulated from its surroundings.
As \(Q=0\) the energy equation for an adiabatic process in a closed system reduces to:
\(-W=(u_2-u_1)\)
In common with the constant volume process this relation applies to reversible and irreversible adiabatic processes as it depends only on the end states and not the path between states.
Unlike processes previously considered there is no straightforward function expressing work for a reversible adiabatic process between two equilibrium states in terms of \(p\) and \(v\). If an algebraic relationship of the form \(u = f(p,v)\) can be established and \(p_1\), \(p_2\), \(v_1\) are known, \(v_2\) can be derived and the work determined.
A practical method of determining work associated with a reversible adiabatic process uses the property entropy, the subject of future tutorials
Isothermal process
A process of expansion or compression in a closed system is called isothermal when the temperature remains constant in the mass of fluid for all combinations of pressure and volume throughout the process. Because a true isothermal process precludes temperature gradients in the mass of fluid the process is inherently reversible. However the term is sometimes applied to a process where the average temperature of the fluid is constant.
In common with a reversible adiabatic process, the work associated with an isothermal expansion or compression can be determined from an algebraic relationship of the form \({\theta}=f(p,v)\)* or more simply from a table of fluid properties that includes entropy.
(* \({\theta}\) denotes an arbitrary temperature scale. We explain thermodynamic temperature scales in a future tutorial).