Familiarity with the previous tutorial covering thermodynamic properties of liquids and vapours is recommended.
Definition of a perfect gas, Equation of state for perfect gases, Definitions of specific heats \(c_p\) and \(c_v\)and their relationship with gas constant R, Expressions for internal energy, enthalpy and entropy in terms of measurable properties (pressure, temperature and volume).
This tutorial outlines the principal thermodynamic properties of gases which, at pressures and temperatures generally experienced in engineering applications, behave as perfect gases. Such applications include gas turbines, reciprocating internal combustion engines, gas compressors, fans and blowers.
Definition of a perfect gas
In the previous tutorial we illustrated properties of vapours, namely gases in a state close to or in transition between liquid and gas phases, using a diagram of pressure \(p\) against specific volume \(v\). Figure 1* is a similar diagram for nitrogen (N2), the major constituent of air. The diagram includes selected isotherms.
* source - wikimedia commons licence
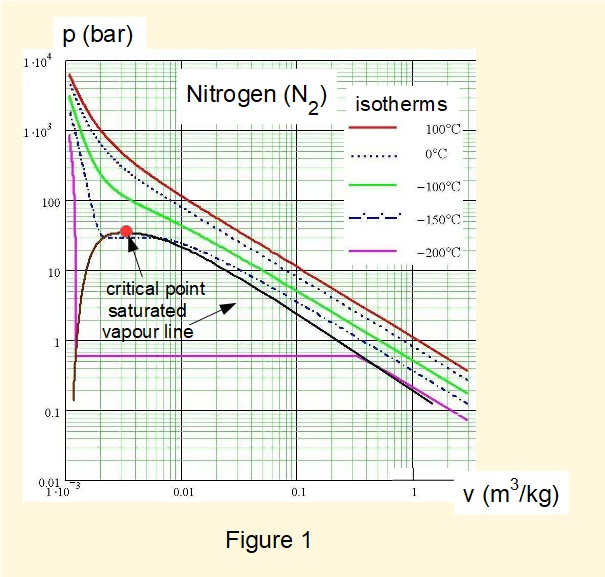
Recall that liquid and gas phases are in equilibrium at the critical point which in the case of nitrogen occurs at pressure 34.0 bar and temperature 126.2 °K (-147.0 °C). Nitrogen gas cannot condense to liquid at temperatures above 126.2 °K, indicated by the fact that all isotherms for temperatures above the critical temperature do not intersect the saturated vapour line. The critical temperature of oxygen, the other main constituent of air, is 154.4 °K.
For practical purposes we consider gases with absolute temperature significantly below ambient temperature to be perfect gases, often called permanent gases although the perfect gas model becomes less valid at pressures from around 10 bar and upwards, depending on the gas. Superheated steam at low pressures, which we conventionally call water vapour, also behaves as a perfect gas.
Equations of state for perfect gases
The basic equation of state for perfect gases expresses the classic gas laws (Boyle's and Charles's Laws), namely:
\(pv=RT\) where \(R\) is the specific gas constant unique to each gas
units: p (kPa) v \(\large(\frac{m^3}{kg})\) T (°K) R \(\large(\frac{kJ}{kg\degree{K}})\)
The specific gas constant \(R=0.287 \large\frac{kJ}{kg\degree{K}}\) is normally used for calculations where air is the working fluid.
For known mass \(m(kg) \) of a particular gas the above equation becomes: \(pV=mRT\) with volume V expressed in \(m^3\).
Avogadro's Law states that the volume \(v_0\) of 1 mole* of all perfect gases is the same when measured at the same pressure and temperature**. This fact leads to the universal equation of state:
\(pv_0=R_0T\) where \(R_0=8.3143 \large\frac{kJ}{kg-mol\degree{K}}\) is the universal gas constant
At standard atmospheric pressure and temperature \( v_0=22.41 \; \large\frac{m^3}{kg-mol}\)
For volume \(V (m^3)\) of gas containing \(n\) moles this equation becomes: \(pV=nR_0T\)
* one mole is the molecular weight of a substance expressed in mass units. In engineering applications the favoured unit is kg-mol. In general chemistry the standard unit is gram-mol. One gram-mol of gas contains the number of molecules defined by Avogadro's number = \(\mathrm{6.023 \; x \; 10^{23}}\)
Specific heats \(c_v\) and \(c_p\)
In broad terms specific heat is the amount of heat \(Q\) required to raise the temperature of unit mass of a substance by one degree. The amount of heat required depends on the conditions under which heat is supplied.
In thermodynamics there are two definitions of specific heat:
- specific heat at constant volume denoted by \(\bm{c_v}\) based on the amount of heat required during a reversible constant volume process.
- specific heat at constant pressure denoted by \(\bm{c_p}\) based on the amount of heat required during a reversible constant pressure process.
specific heat at constant volume is expressed as:
\(\dfrac{Q_v}{\Delta T}\) where \(Q_v\) is the the quantity of heat supplied during a reversible constant volume process.
In a previous tutorial we established for a reversible constant volume process that: \(dQ=du\)
Thus we can define \(\bm c_v = \Large(\tfrac{du}{dT})\normalsize{_v}\) subscript \(v\) denoting constant volume
specific heat at constant pressure is expressed as:
\(\dfrac{Q_p}{\Delta T}\) where \(Q_p\) is the the quantity of heat supplied during a reversible constant pressure process.
In a previous tutorial we established for a reversible constant pressure process that: \(dQ=dh\)
Thus we can define \(\bm c_p = \Large(\tfrac{dh}{dT})\normalsize{_p}\) subscript \(p\) denoting constant pressure
Units for \(c_v\) and \(c_p\) are \(\large\frac{kJ}{kg\degree{K}}\). Values for specific substances are derived experimentally.
\(c_v\) and \(c_p\) prove to be very useful when deriving expressions for properties \(u\), \(h\) and \(s\) for a perfect gas, as now shown.
Derived expressions for \(u\), \(h\) and \(s\)
For processes where the working fluid is a perfect gas \(p\) and \(T\) can generally be measured and then \(v\) determined from the equation of state: \(pv=RT\). We now outline steps leading to expressions for \(s\), \(h\) and \(u\) in terms of the measurable variables.
internal energy \(u\)
From the premise that for perfect gas \(u\) is a function only of temperature \(T\) (known as Joule's Law) the following can be deduced:
- \(c_v\) must also be a function only of temperature \(T\) (or constant)
- the definition of \(c_v\) applies to any infinitesimal process and is not restricted to a constant volume process.
We can now write the definition of \(c_v\) as: \( c_v = \Large(\tfrac{du}{dT})\) or: \(du=c_v.dT\) -------- (1)
For pressures and temperatures found in most practical applications \(c_v\) is considered to be constant. Thus \(c_v=constant\) is a defined property of a perfect gas.
Integrating equation (1) between states 1 and 2 gives: \(u_2-u_1 = c_v(T_2-T_1)\) -------- (2)
enthalpy \(h\)
By definition: \(h=u+pv\)
Applying \((pv=RT)\) gives: \(h=u+RT\) -------- (3)
Now substitute \(du\) from equation (1) into equation (3) giving:
\(dh= c_v.dT+R.dT\) and hence \(dh=(c_v+R).dT\) -------- (4)
showing that for a perfect gas \(h\) is also a function of temperature alone.
By a similar justification that removed the restriction of a constant volume process from the definition of \(c_v\) we apply the same criterion to the definition of \(c_p\) regarding a constant pressure process giving:
\( c_p = \Large(\tfrac{dh}{dT})\) or: \(dh=c_p.dT\) -------- (5)
Now substitute \(dh\) from equation (4) into equation (5) giving:
\((c_v+R).dT=c_p.dT\) from which it follows that \(c_p-c_v=R\) -------- (6)
Integrating equation (5) between states 1 and 2 gives: \(h_2-h_1 = c_p(T_2-T_1)\) -------- (7)
From equation (6) it can be seen that for a perfect gas \(c_p\) must be constant, having previously determined that \(c_v\) and \(R\) are constant*. It also follows that \(c_p > c_v\) and the ratio designated \(\gamma (gamma)=\dfrac{c_p}{c_v}\) is constant.
The following values are generally used in calculations, based on air at temperature \(300\degree K\):
\(c_p=1.005 \large\frac{kJ}{kg\degree K}\) \(c_v=0.718 \large\frac{kJ}{kg\degree K}\) \(\gamma=1.4\)
* a gas that follows the gas law \(pV=nR_0T\) but has specific heats \(c_v\) and \(c_p\) that vary with temperature is called an ideal gas.
entropy \(s\)
Consider two fundamental equations in differential form from previous tutorials:
\((dQ)_{rev}=du+p.dv\) which is a statement of the First Law of Thermodynamics.
\((dQ)_{rev}=T.ds\) which defines entropy as a consequence of the Second Law of Thermodynamics.
Combining these equations gives: \(T.ds=du+p.dv\)
Giving: \(ds=\dfrac{1}{T}.du+\dfrac{p}{T}.dv\) -------- (8)
Applying \((pv=RT)\) to equation (8) gives: \(ds=\dfrac{1}{T}.du+\dfrac{R}{v}.dv\)
and substituting \((du=c_v.dT\)) from equation (1) where \(c_v\) is constant gives:
\(ds=\dfrac{c_v}{T}.dT+\dfrac{R}{v}.dv\) -------- (9)
Integrating equation (9) between states 1 and 2 gives: \(s_2-s_1=\large\int_1^2 \normalsize\dfrac{c_v}{T}.dT+\large\int_1^2 \normalsize\dfrac{R}{v}.dv\)
giving: \(s_2-s_1=c_v.ln\dfrac{T_2}{T_1}+R.ln\dfrac{v_2}{v_1}\) -------- (10)
Equation (10) shows that entropy of a perfect gas is a function of two properties, in this case \(T\) and \(v\). Using the equation of state (\(pv=RT)\) and the relation \((c_p-c_v=R)\) expressions for other pairs of measurable properties are easily derived from equation (10), namely:
\(s_2-s_1=c_v.ln\dfrac{T_2}{T_1}-R.ln\dfrac{p_2}{p_1}\) -------- (11)
\(s_2-s_1=c_v.ln\dfrac{p_2}{p_1}+c_p.ln\dfrac{v_2}{v_1}\) -------- ( 12)
Note that equations (2), (7), (10), (11) and (12) are valid between any two states regardless of the process between state 1 and state 2.
Next: Thermodynamics contents
I welcome feedback at: