In previous tutorials we applied the First Law of Thermodynamics to processes for:
In this tutorial we show how such processes combine to create thermodynamic cycles. We consider only closed cycles where the working fluid is contained within the system and its properties are identical at any arbitrary start and end point of the cycle.
The term open cycle typically describes any system where the working fluid enters from and exits to the surroundings and can comprise one or any number of linked items. Study of open cycles is based on the steady-flow energy equation.
Closed cycles comprise linked sequences of steady-flow processes or closed processes.
We consider the following closed cycles:
*in thermodynamics a heat engine is any system that converts heat into work
Cycles (1) and (2) are vapour cycles where the fluid alternates between liquid and vapour phases during the cycle. In cycle (3) air is the notional working fluid*.
*for an introduction to properties of fluids refer to tutorials covering vapours and perfect gases

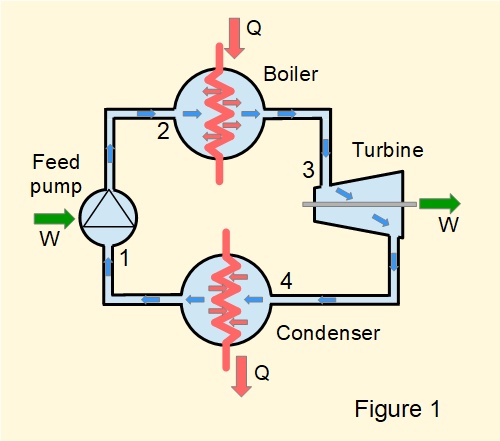
Figure 1 illustrates a closed vapour cycle for power generation linking four items of plant with water/steam as the working fluid. This is known as the Rankine cycle.
Referring to numbered points on the diagram, stages in the cycle are as follows.
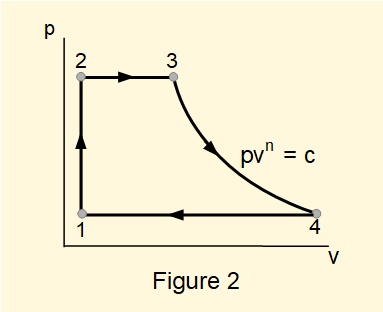
In a previous tutorial we introduced pressure/volume \((pv)\) diagrams showing changes in thermodynamic properties of an element of fluid throughout a process. \(pv\) diagrams for complete cycles are constructed by linking \(pv\) plots for each stage of the cycle. For convenience all processes are illustrated as being reversible with solid lines linking end states.
Figure 2 shows the stages of a Rankine cycle on a \(pv\) diagram.
Steady-flow energy equations for each stage of the Rankine cycle derived in a previous tutorial for absolute values of \(Q\) and \(W\) are as follows (in units kJ/kg):
giving for the complete cycle:
\(Q_{net}=(Q_{23}-Q_{41})=(h_3-h_2-h_4+h_1)\) .
\(W_{net}=(-W_{12}+W_{34})=(-h_2+h_1+h_3-h_4)\)
thus for the complete cycle:
\(Q_{net}=W_{net}\) and \(Q_{net}-W_{net}=0\)
This result for a closed cycle is consistent with the First Law of Thermodynamics.
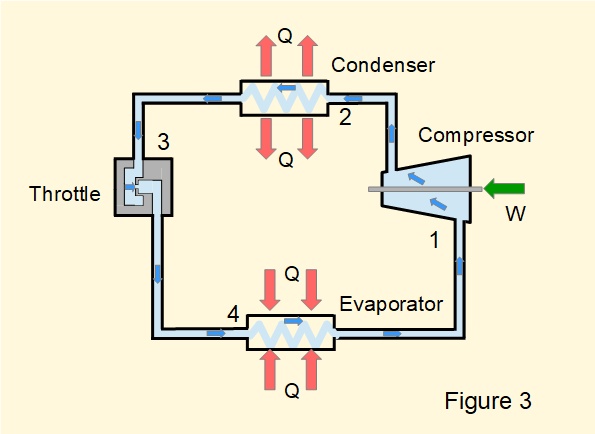
Figure 3 illustrates a closed vapour cycle for a refrigerator or heat pump. Refrigeration cycles use a variety of working fluids, known as refrigerants selected for physical properties compatible with the range of external temperatures prevailing at the evaporator and condenser.
The refrigeration cycle is almost a reversed version of the Rankine cycle. Referring to numbered points on the diagram, stages in the cycle are:
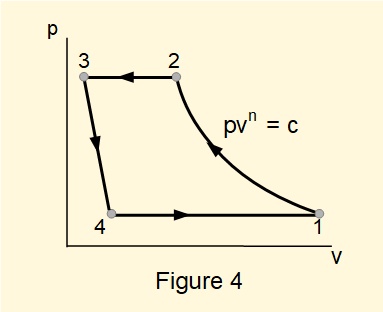
Figure 4 shows a \(pv\) diagram for a refrigeration cycle summarised as follows:
Steady-flow energy equations for each stage of the refrigeration cycle derived in a previous tutorial for absolute values of \(Q\) and \(W\) are (in units kJ/kg):
giving for the complete cycle:
\(Q_{net}=(Q_{41}-Q_{23})=(h_1-h_4-h_2+h_3)=(h_1-h_2)\) .
\(W_{net}=-W_{12}=(h_1-h_2)\)
thus for the complete cycle:
\(Q_{net}=W_{net}\) and \(Q_{net}-W_{net}=0\)
Again, this result for a closed cycle is consistent with the First Law of Thermodynamics.
In a previous tutorial we used the Otto cycle as an example of the First Law applied to a cycle of closed processes, illustrating for a complete cycle that:
\((Q_{net}-W_{net})=0\)
Here we show another cycle of closed processes*, known as the Carnot cycle, which has very useful properties for developing the Second Law of Thermodynamics. as explained in the next tutorial. We illustrate the Carnot cycle in in Figures 5 and 6 with air as the notional working fluid.
(*The Carnot cycle can also be envisaged as a closed cycle containing linked open-flow elements, similar to the Rankine cycle above)
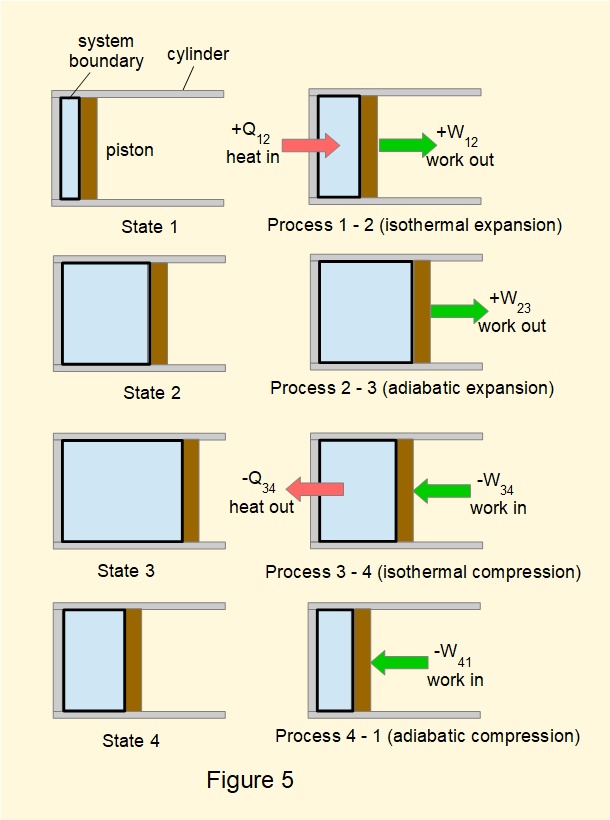
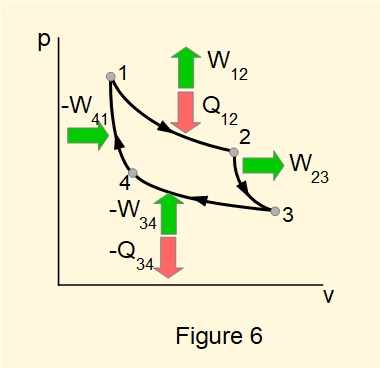
A key feature of the Carnot cycle is that heat input \(Q_{12}\) and heat output \(Q_{34}\) are isothermal processes, that is they take place at a constant temperature.
By the First Law: \(Q_{net}=W_{net}\)
giving: \(Q_{12}-Q_{34}=(W_{12}+W_{23})-(W_{34}+W_{41})\)
which in general terms for any heat engine can be stated as: \(Q_{in}-Q_{out}=W_{net}\)
Next: The Second Law of Thermodynamics
I welcome feedback at: